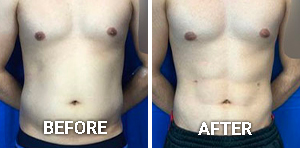
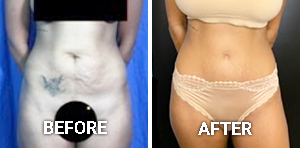
Facial Rejuvenation in Beverly Hills

Facial rejuvenation is a suite of surgical and non-surgical treatments to restore a radiant appearance, reduce the common signs of aging, and bring out a younger, more confident you! By addressing wrinkles, sunspots, volume loss, and other aesthetic blemishes, Dr. Michael Omidi MD, FACS, and his Beverly Hills team can […]
Read More